Nitric oxide (NO) is one of the most important, and often underestimated, gases in the field of air qualityAir quality refers to the state of the air we breathe and its composition in terms of pollutants present in the atmosphere. It is considered good when poll...
Read more and industrial emissions management. Colourless, odourless and highly reactive, NO belongs to the group of nitrogen oxides (NOx), compounds directly linked to air pollution, photochemical smogSmog, beyond that dense fog
Smog is a mixture of air pollutantsAir pollution caused by atmospheric contaminants is one of the most critical and complex environmental problems we face today, both because of its global r...
Read more that accumulate in the atmosphere, especially in urban areas. This phenomenon is character...
Read more and the deterioration of respiratory health.
Understanding what nitric oxide is, what its sources in the air are and how it is measured is the starting point for any environmental and industrial monitoring strategy.
In 2017, one of the worst years on record for air quality in Madrid, specifically in October, Madrid City Council activated the high pollution protocol after exceeding the nitrogen dioxide (NO2)Nitrogen dioxide (NO2) is a harmful gas whose presence in the atmosphere is mainly due to the use of fossil fuels in combustion vehicles and industrial act...
Read more limit, reducing the speed limit on the M-30 and restricting access roads to the city to 70 km/h. It was not an isolated episode. That same year, 15 of the city’s 24 air quality monitoringControlling air quality is an essential task in order to enjoy optimal environmental conditions for healthy human development and to keep the environment i...
Read more stations exceeded the annual NO2 limit set at 40 µg/m3 by the European directive and at least 7 stations exceeded the hourly limit value of 200 µg/m3 on more than 18 occasions, breaching EU legislation for the eighth consecutive year.
Behind those readings was nitric oxide emitted on a massive scale by road traffic. NO oxidises rapidly in the urban atmosphere and is transformed into NO2, the pollutant ultimately recorded by air quality monitoring stations.
Although NO has a short atmospheric lifetime, this direct and dangerous cycle for air quality makes real-time nitric oxide monitoring essential. It is an operational requirement, both for urban authorities and for industrial facilities subject to emissions regulations.
Air Quality Innovation in Just 1 Click
Stay informed about the air you breathe!
Subscribe to our newsletter to receive the latest updates on environmental monitoring technology, air quality studies, and more.
Throughout this article, we will analyse the main sources of nitric oxide in the air (from combustion in engines and industrial processes to diffuse emissions), its relationship with NOx and its effects on health and the environment. We will also review the nitric oxide sensor technologies currently available and the keys to designing effective measurement systems, even for the most demanding environments.

Nitric oxide monitoring is the link that connects regulation with reality.
What is nitric oxide (NO)?
Nitric oxide is an inorganic gas made up of one nitrogen atom and one oxygen atom, with the molecular formula NO, a simple molecule that might invite people to downplay it compared with other industrial pollutants, yet it is so reactive that it can alter the entire chemistry of the troposphere.
Its molecular weight (30.01 g/mol) makes it surprisingly light, almost identical to the weight of the air we breathe. This is why it does not move through the atmosphere under the effect of gravity, but instead mixes and disperses easily, both outdoors and indoors. Thanks to its high reactivity, when NO is present in atmospheric oxygen, it oxidises rapidly and becomes NO2, triggering a chain of reactions with direct consequences for air quality.
NO vs. NO2: two gases, one common origin
Although they are often mentioned together, NO and NO2 are different compounds with distinct behaviours and effects:
| Property | Nitric oxide (NO) | Nitrogen dioxide (NO2) |
| Colour | Colourless | Reddish-brown |
| Odour | Odourless | Pungent, irritating |
| Water solubility | Very low | Moderate |
| Direct toxicity | Medium | High |
| Atmospheric lifetime | Seconds to minutes | Hours |
| Predominant origin | Direct combustion | Oxidation of NO |
NO is the gas emitted in the highest proportion during combustion processes (engines, boilers and industrial furnaces), whereas NO2 is largely considered a secondary pollutant. It forms when NO reacts with ozone (O3) or oxygen (O2)Oxygen (O2) is the most critical safety parameter to measure in industrial environments involving confined spaces, combustion processes, biogas generation ...
Read more in the air. This distinction is essential to understand why measuring NO at the emission source is more revealing than waiting to detect the NO2 into which it has been transformed.
In the field of air pollution, the term nitrogen oxides (NOx) covers all gaseous compounds formed by the combination of nitrogen and oxygen. When approached from the perspective of emissions regulation, however, the term NOx refers almost always to the sum of NO + NO2, the two compounds that are most abundant and most regulated in the atmosphere.
Both European industrial emissions legislation (the IED Directive) and ambient air quality standards (Directive 2008/50/EC) set limit values precisely in terms of NO2 equivalent, which makes NO the precursor that every industrial facility should control at source.
Nitric oxide in atmospheric chemistry
NO plays a fundamental role in tropospheric chemistry, acting as a catalyst for the formation of ground-level ozone and as a precursor of photochemical smog. The chemical cycle involved is as follows:
- Combustion emits NO into the atmosphere.
- NO reacts with tropospheric ozone (O3). This process consumes ozone but generates NO2.
- Solar radiation causes photodissociation of NO2, releasing atomic oxygen.
- Atomic oxygen reacts with O2, generating ozone.
- In the presence of volatile organic compounds (VOCs), the cycle becomes unbalanced and ozone accumulates, forming photochemical smog.
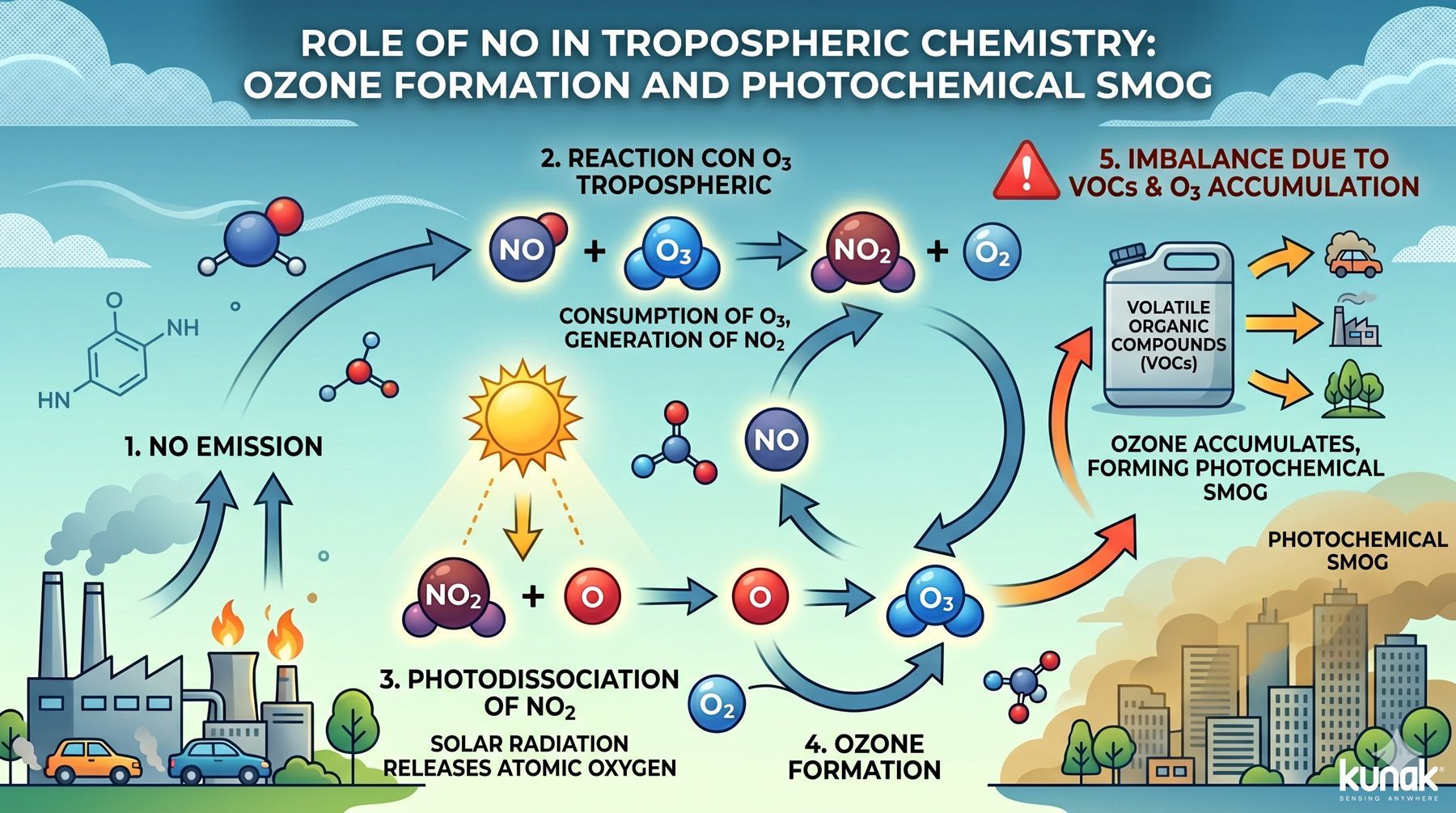
Role of nitric oxide in tropospheric chemistry: ozone formation and photochemical smog
This process explains why urban areas with heavy traffic or high industrial density experience ozone peaks during hours of maximum sunlight. It is also the way to approach control of nitric oxide emissions as a direct lever for improving air quality at urban scale.
Sources of nitric oxide emissions in the atmosphere
Nitric oxide emissions have a dual origin. While human activity is the main contributor to a large proportion of NO present in urban and industrial environments, natural processes contribute diffusely but continuously to the overall balance of the gas in the atmosphere. Understanding both sources of nitric oxide emissions is essential for designing effective monitoring and regulatory compliance strategies.
Anthropogenic sources
Human-related sources are the most significant due to their local impact and the importance of regulatory control. In general, they originate from high-temperature combustion, when nitrogen and oxygen in the air are heated above 1,200 °C and react to form NO. This process is known as thermal NO formation or the Zeldovich mechanism.
The hotter an engine burns, the more nitric oxide it produces. This is not because the fuel is dirty, but because the air itself transforms under extreme heat.
This is equivalent to saying that air breaks down under heat. Oxygen and nitrogen are gases that naturally coexist in the atmosphere without reacting with each other. They are not by-products, nor even gases from the combustion process itself, but components of the air that remain inert until the extreme temperature applied (diesel engine, industrial boiler or gas turbine) triggers an exceptional reaction. Oxygen and nitrogen molecules break apart, generating highly reactive atoms that cannot remain isolated for long and therefore combine immediately to form nitric oxide.
Road traffic and maritime transport
Road traffic is the main anthropogenic source of NO in urban environments, especially diesel engines, which operate with excess air and high combustion temperatures, ideal conditions for NO formation. Engines in cargo ships and cruise vessels represent a growing source of NOx emissions in port areas and along coastal routes. The International Maritime Organization (IMO) established Tier III regulations setting NOx limits for new ships operating in Emission Control Areas (ECAs), achieving reductions of up to 80% compared to previous levels.
Thermal power plants and industrial combustion
Power generation plants using coal, natural gas or fuel oil are major point sources of NOx. In the EU, these facilities are subject to Emission Limit Values (ELVs) set by the Best Available Techniques Reference Documents (BREF) derived from the Industrial Emissions Directive (IED). For large combustion plants, current NOx limits typically range between 50 and 200 mg/Nm3 depending on fuel type and installed capacity, making continuous monitoring mandatory through CEMS (Continuous Emission Monitoring Systems).
Refineries, petrochemicals and the oil & gas sector
In the oil & gas sector, NO emissions originate from multiple sources such as process heaters, boilers, gas turbines, flares and, to a lesser extent, fugitive emissions from combustion equipment. Refineries are particularly complex facilities because they combine point-source emissions with diffuse sources distributed across the site. The refinery BREF and the EPA 40 CFR Part 60 standard in the United States establish specific NOx measurement and reporting protocols that include NO as the dominant fraction.

Agricultural and forest soils are a continuous source of NO due to nitrification and denitrification processes carried out by soil bacteria.
Natural sources
Although natural sources of NO have less local impact than anthropogenic ones, they contribute significantly to the global nitrogen cycle and influence NO levels in areas far from industrial activity. The main natural sources of NO are:
Soil microbial activity
Agricultural and forest soils are a continuous source of NO due to nitrification and denitrification processes carried out by soil bacteria. During nitrification, bacteria oxidise ammonium (NH4) to nitrate (NO3), releasing NO as a by-product. The intensive use of nitrogen fertilisers amplifies this process, turning agricultural soils into a diffuse source of emissions that is difficult to quantify and control.
Lightning discharges
Lightning generates instantaneous temperatures of up to 29,700 °C within the lightning channel, sufficient to break N2 and O2 molecules in the air and directly produce NO. Thunderstorms are estimated to generate between 2 and 8 Tg of nitrogen in the form of NOx per year globally, a smaller proportion than anthropogenic sources but with a relevant role in free tropospheric chemistry and in the lower stratosphere.
Once in the free troposphere (the region of the atmosphere starting approximately 1–2 km above ground level, beyond the direct influence of the Earth’s surface), NOx and its derivatives no longer disperse locally but travel with air masses, potentially reaching areas hundreds of kilometres away from the original emission source.
Wildfires
Biomass combustion during wildfires releases large amounts of NO along with CO, particles and volatile organic compounds. Wildfires act as mobile and intermittent sources of high intensity. Smoke plumes can transport NO and its derivatives over hundreds of kilometres from the original source, temporarily increasing tropospheric ozone levels in geographically distant areas.
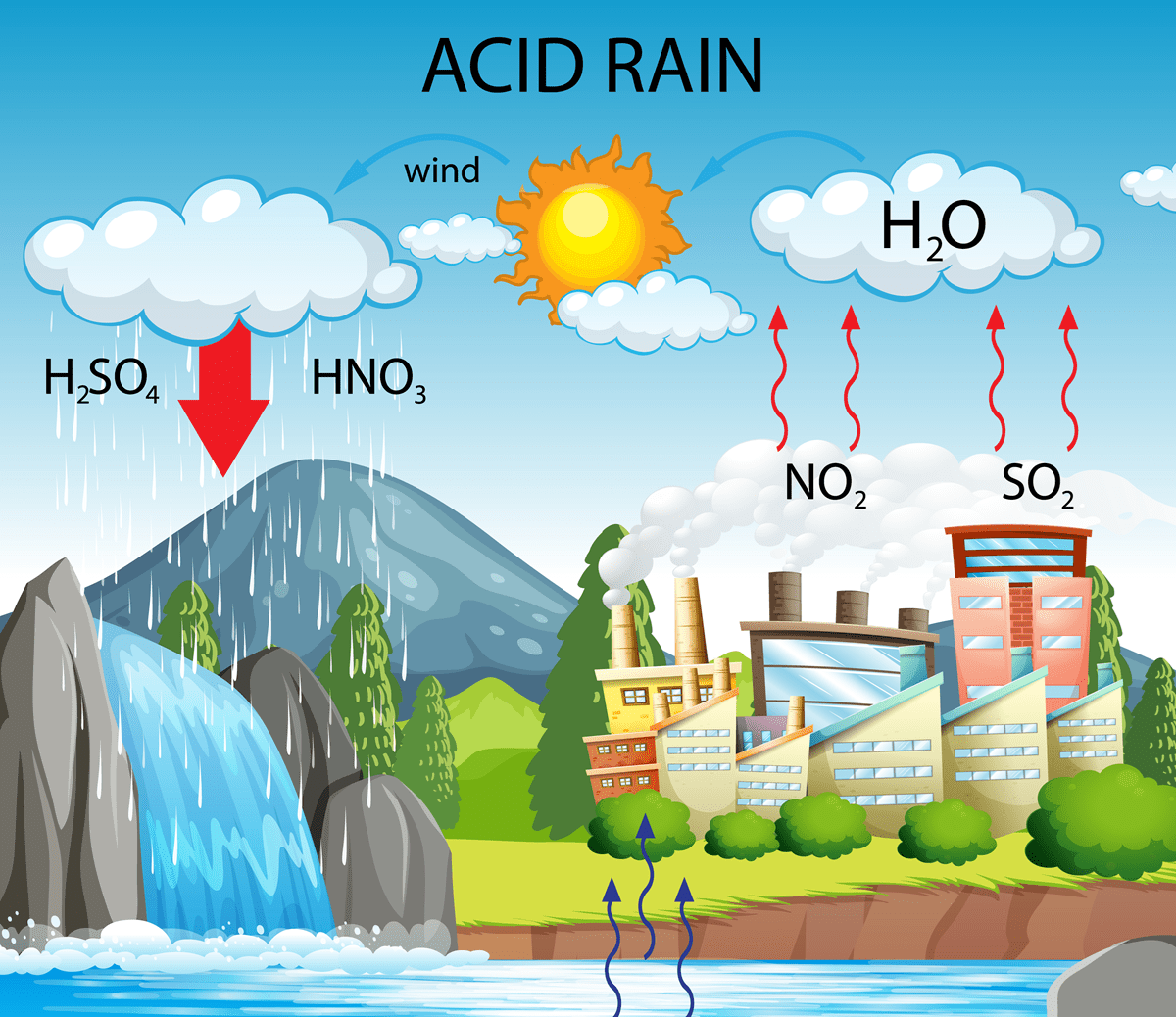
NO2 reacts with water vapour to form nitric acid (HNO3), causing acid rain and damaging ecosystems and materials.
Impact of nitric oxide on air quality and health
The impact of nitric oxide on air quality goes beyond its direct presence as a primary pollutant. NO acts as a trigger for a cascade of environmental and health effects that affect both ecosystems and human health, with a particularly strong impact on urban populations and industrial environments chronically exposed to its presence.
Environmental effects
The presence of NOx in urban and industrial atmospheres degrades air quality both directly and indirectly. NO2, formed from NO, is one of the most widely monitored pollutants in Europe. Its ability to reduce visibility is evident (responsible for the characteristic brown haze over large cities on calm days), but this is only the most visible symptom of pollution that reaches dangerous concentrations at the molecular level.
94% of the European urban population remains exposed to concentrations of fine particles (PM2.5) that exceed WHO guidelines, pollution to which NOx directly contributes as a precursor to the formation of secondary particles in the atmosphere. European Environment Agency (EEA). Air quality status report 2025.
In addition to NO’s role in the formation of tropospheric ozone, NOx contributes to two large-scale environmental phenomena:
- Acid rain: NO2 reacts with atmospheric water vapour to form nitric acid (HNO3), which precipitates with rainfall and acidifies soils and water bodies, damaging forest and aquatic ecosystems and deteriorating construction materials and cultural heritage.
- Eutrophication: nitrogen deposited by NOx emissions acts as a nutrient in aquatic and terrestrial ecosystems, disrupting species balance and promoting excessive algal growth in lakes and coastal areas.
These effects extend beyond the emission source through atmospheric transport. NOx and its derivatives can travel hundreds of kilometres from their origin, turning local industrial and urban emissions into a regional and transboundary issue.

The rapid conversion of NO into NO2 and its role in the formation of tropospheric ozone make it an indirect precursor of serious health effects.
Effects on human health
NO itself has relatively moderate direct toxicity at typical ambient air concentrations. However, its rapid conversion into NO2 and its role in the formation of tropospheric ozone make it an indirect precursor of serious health effects, including:
Respiratory irritation and acute effects
NO2, the main derivative of NO in urban atmospheres, is a powerful irritant to the respiratory mucosa. Exposure to high concentrations causes inflammation of the airways, increased bronchial reactivity and greater susceptibility to respiratory infections.
Tropospheric ozone, generated with the involvement of NO, further aggravates these effects. At concentrations above 100 µg/m3 (thresholds frequently exceeded in summer in large Mediterranean cities), it causes coughing, reduced lung function and asthma exacerbation. The most vulnerable groups are children, older adults and individuals with pre-existing respiratory or cardiovascular conditions.
Cardiovascular and chronic respiratory diseases
Chronic exposure to NO2 is directly linked to the development of long-term diseases. In 2021, the WHO revised its air quality guidelines and reduced the annual NO2 guideline value from 40 µg/m3 to 10 µg/m3, as large-scale epidemiological studies demonstrated health effects at much lower concentrations than those set in current European legislation. Documented effects include:
- Chronic obstructive pulmonary disease (COPD): prolonged exposure to NOx accelerates the decline in lung function, particularly in individuals with occupational exposure in industrial environments.
- Ischaemic heart disease: systemic inflammation induced by oxidising pollutants such as NO2 and O3 is associated with an increased risk of myocardial infarction and stroke.
- Lung development in children: cohort studies in high-traffic areas show that children chronically exposed to elevated NO2 levels develop reduced lung capacity, with effects that may be irreversible in adulthood.
The health burden of NOx is, in both economic and human terms, one of the highest among all regulated air pollutants. According to the EEA (2025), exposure to NO2 causes around 34,200 premature deaths per year in Europe, reinforcing the need for nitric oxide monitoring systems that are accurate, continuous and strategically deployed in both urban and industrial environments.
Regulatory framework and international guidelines on NOx
The regulation of NOx emissions and their derivatives operates at multiple levels simultaneously, including ambient air quality, industrial emissions and mobile sources. Understanding how these regulatory frameworks interact is essential for any environmental compliance manager, since nitric oxide (although not always directly regulated) is the precursor of NO2, the pollutant that is subject to specific legal limits.
The Directive 2008/50/EC of the European Parliament and of the Council on ambient air quality and cleaner air for Europe is the reference framework for EU Member States regarding the assessment and management of NO2. It establishes two binding limit values for NO2 in ambient air:
- Annual limit value: 40 µg/m3 (annual mean).
- Hourly limit value: 200 µg/m3, not to be exceeded more than 18 times per calendar year.
A key update to this framework was introduced in October 2024 with the publication of Directive (EU) 2024/2881, a recast of the air quality directive that introduces stricter limits aligned with the 2021 WHO guidelines and must be transposed into national legislation by Member States before 2030.
The new proposed annual NO2 limit value in this revision is reduced to 20 µg/m3, an intermediate step towards the 10 µg/m3 recommended by the WHO.
In September 2021, the World Health Organization published an update of its Global Air Quality Guidelines. For NO2, it established the following guideline levels:
| Averaging period | WHO guideline 2005 | WHO guideline 2021 |
| Annual | 40 µg/m3 | 10 µg/m3 |
| 24-hour | — | 25 µg/m3 |
The reduction to 10 µg/m3 annually (four times stricter than the current EU legal limit) reflects the accumulated scientific evidence on the health effects of NO2 at low concentrations, especially under chronic exposure. These guidelines are not legally binding, but they serve as the technical benchmark guiding regulatory revisions worldwide.
In the United States, the Environmental Protection Agency (EPA) regulates NO2 through the National Ambient Air Quality Standards (NAAQS). These set an annual limit value of 100 µg/m3 (0.053 ppm) for NO2 in ambient air (more permissive than the European standard) and an hourly standard of 188 µg/m3.
For industrial emissions, the key instrument is 40 CFR Part 60 (New Source Performance Standards), which establishes NOx limits for new stationary sources across sectors such as power plants, refineries, gas turbines and other industrial facilities.
The relationship between NO, NO2 and regulatory limits
The key principle underlying the entire regulatory framework is that legislation regulates NO2, but the source of the problem is NO. This approach is based on technical and practical reasons:
- NO2 is the measurable and accumulative pollutant in ambient air that offers greater technical feasibility.
- NO is converted into NO2 in the atmosphere within minutes or hours, so controlling NO at source is equivalent to preventing NO2 at the receptor.
- Industrial emissions regulations (IED/BREF, 40 CFR Part 60) control total NOx at source, including NO as the main fraction, while ambient air quality regulations set limits on NO2 as an indicator of actual health impact.
This dual regulation (at source and at receptor) justifies the need for continuous nitric oxide monitoring systems both in industrial stacks (CEMS) and in urban air quality monitoring networksAir quality monitoring networks consist of an organised system of monitoring stations distributed across various locations to measure and assess air pollut...
Read more.

Measuring NO and NO2 is one of the pillars of smart city platforms focused on air quality management.
Monitoring of nitric oxide
Nitric oxide monitoring is the link that connects regulation with reality. Without continuous and accurate measurement of NO and its derivatives, public authorities cannot manage air quality and industrial facilities cannot demonstrate compliance with current regulations. Available technologies range from reference analysers used in official networks to low-cost sensors integrated into dense urban monitoring networks.
Reference methods for NO measurement
The chemiluminescence method is the reference method for measuring NO and NO2 in ambient air, established by the European standard UNE-EN 14211 and equivalent to the EPA reference method (40 CFR Part 50, Appendix F). Its operating principle is based on the reaction between NO in the air sample and ozone generated internally in the instrument:
NO + O3 → NO2* + O2
The excited NO2 molecule (NO2*) emits photons as it returns to its ground state, and this light emission (proportional to NO concentration) is detected by a highly sensitive photomultiplier. The result is extremely precise measurement, with typical detection limits of 0.4 to 1 ppb (parts per billion).
To measure NO2, the analyser includes a molybdenum converter (or photolytic in more advanced equipment) that first converts NO2 into NO before detection, obtaining the total NOx concentration; the difference between NOx and NO yields NO2. Chemiluminescence analysers are used in air quality monitoring networks operated by regional authorities in Spain and in national networks across the EU.
Their main limitations are high cost (between €10,000 and €25,000 per unit), the need for periodic calibration and maintenance requirements. Together, these factors make large-scale deployment unfeasible, which has driven the development of alternative technologies.
Nitric oxide sensors and emerging technologies
Electrochemical NO sensors operate by measuring the current generated by the electrochemical oxidation of the gas in a three-electrode cell. They are compact, low power and cost-effective, making them the dominant technology in portable applications, wearable devices for occupational exposure and low-cost monitoring networks. Their key characteristics include:
- Measurement range: typically 0–250 ppm in industrial applications, 0–5 ppm in air quality.
- Response time: 15–30 seconds (T90).
Their main limitation is cross-sensitivity to other gases (NO2, CO, H2S), as well as humidity and temperature, which require compensation algorithms or field calibration, as implemented by Kunak.
Applications of NO monitoring
In urban environments, NO monitoring fulfils three simultaneous functions: feeding early warning systems that trigger traffic restriction protocols, assessing the effectiveness of measures such as Low Emission Zones (LEZs) and providing population exposure data that underpin public health policies.
In the industrial sector, continuous NOx monitoring is both a legal requirement and a process optimisation tool. In refineries and petrochemical plants, CEMS installed in process heaters, boilers and turbines allow real-time adjustment of combustion parameters (excess air, flame temperature, flue gas recirculation) to minimise thermal NO formation without compromising energy efficiency. In cogeneration plants and gas facilities, integrating CEMS data with DCS control systems enables adaptive management of low-NOx burners.
Without robust monitoring data, environmental policy operates blindly.
Applications of nitric oxide across sectors
Nitric oxide, beyond being a pollutant that must be measured and controlled, is, due to its reactivity and physicochemical properties, a highly useful compound in industrial, environmental and biomedical applications.
Industry and emissions control
In the industrial sector, precise knowledge of NO behaviour is the foundation of the most widely used NOx emission reduction technologies worldwide:
Selective catalytic reduction (SCR)
SCR technology is the standard for NOx control in thermal power plants, large industrial boilers and heavy-duty vehicle engines. Its principle consists of injecting urea or ammonia (NH3) into the flue gas stream, where it reacts with NO in the presence of a vanadium-titanium catalyst to produce nitrogen (N2) and water vapour.
NOx reduction efficiencies with SCR reach 80–95%, making it the most effective available technique for high-flow sources. Its proper operation depends on accurate measurement of NO at the inlet and outlet of the catalytic reactor, making NOx CEMS an integral component of SCR systems.
Selective non-catalytic reduction (SNCR)
In installations where catalyst installation is not feasible (due to space, temperature or cost), SNCR injects urea or ammonia directly into the high-temperature combustion zone (850–1,100 °C), achieving NOx reductions of 30–70% without a catalyst. It is commonly used in waste incineration plants and some biomass boilers.
Low-NOx burners
Advanced industrial burner design reduces thermal NO formation by directly acting on flame parameters, such as lowering peak temperature, controlling excess air and recirculating flue gases. Continuous NO monitoring at the outlet provides the feedback signal needed to optimise these parameters in real time.
Environmental monitoring and smart cities
Measurement of NO and NO2 is one of the pillars of smart city platforms focused on air quality management. In this context, NO serves as an early indicator: detecting it at source (before it transforms into NO2 and accumulates) allows earlier and more precise urban management responses.
Main applications in urban environments include:
- Low-cost sensor networks: deployed on streetlights, traffic lights and urban furniture, complementing reference stations with much greater spatial coverage and enabling identification of pollution hotspots in real time.
- Adaptive traffic management: integration of NO data into traffic light control algorithms to redirect traffic flows during high pollution episodes, minimising population exposure in densely populated residential areas.
- Assessment and monitoring of Low Emission Zones (LEZs): NO data before and after LEZ implementation provides objective evidence that allows authorities to justify, refine or expand these measures to citizens and European institutions.
- Air quality digital twins: computational models that integrate real-time NO monitoring data with meteorological and traffic information to predict pollution trends and simulate the impact of different policies before implementation.
Biomedical and scientific applications
The most surprising aspect of nitric oxide is, paradoxically, the one furthest from its image as a pollutant. It plays a role as an essential signalling molecule in the human body, being produced endogenously by the enzyme nitric oxide synthase (NOS). It functions as a messenger in three key physiological systems:
- Cardiovascular system: NO produced by the vascular endothelium acts as a vasodilator, relaxing smooth muscle in blood vessels and regulating blood pressure. This mechanism underpins drugs such as nitroglycerin (used for angina) and sildenafil (the active ingredient in Viagra), which enhances the NO pathway in vascular tissue.
- Immune system: macrophages produce NO at high concentrations as a defence mechanism against bacteria, fungi and tumour cells, leveraging its direct cellular toxicity.
- Nervous system: NO acts as an atypical neurotransmitter (it is not stored in vesicles but produced and diffused instantly) in the modulation of synaptic transmission, memory and neuromuscular coordination.
In the clinical field, measurement of exhaled nitric oxide (FeNO) is an established biomarker for the diagnosis and monitoring of allergic bronchial asthma. Higher NO concentrations in exhaled air indicate eosinophilic airway inflammation, guiding the adjustment of inhaled corticosteroid treatment. This test, performed using portable chemiluminescence analysers, is now a routine clinical tool in pulmonology.

With continuous and precise measurement of NO and its derivatives, public authorities can effectively manage air quality and industrial facilities can demonstrate compliance with current regulations.
Importance of nitric oxide monitoring for environmental management
Nitric oxide cannot be managed if it is not measured. NO monitoring is the point where atmospheric science, environmental regulation, industrial engineering and public policy converge. Without robust real-time data on NO concentration and emissions, air quality management becomes a blind intervention.
Air quality assessment
Measuring NO is essential to understand the real dynamics of air pollution in a given location. While NO2 and ozone are the pollutants perceived by the public and media as air quality indicators, NO is the precursor that determines their evolution. A monitoring network that includes NO (and not only NO2) makes it possible to:
- Identify emission sources with greater precision, distinguishing between road traffic, industrial or natural sources.
- Detect emerging pollution episodes before NO is transformed into NO2 and accumulates in ambient air.
- Characterise the daily and seasonal cycle of NOx pollution, essential information for designing effective management measures.
- Validate atmospheric dispersion models used by authorities to predict episodes and assess emission reduction scenarios.
Spatial and temporal data resolution is therefore critical.
A single reference station per urban district is insufficient to capture the real variability of NO concentrations, which can vary by a factor of ten between a high-traffic street and a park just two hundred metres away.
Regulatory compliance and environmental reporting
For industrial facilities subject to the IED Directive, continuous NOx reporting is not optional. It is a legal obligation and non-compliance may result in penalties, revision of the integrated environmental permit (IEP) or, in severe cases, suspension of operations. Data generated by NOx CEMS must be traceable, verifiable and comparable with the Emission Limit Values (ELVs) set in the permit, and must be reported to the competent authorities at defined intervals, typically including validated hourly averages and daily averages.
Within the framework of the European Pollutant Release and Transfer Register (E-PRTR), large industrial facilities are required to report annually their NOx emissions. This data is integrated into the national emissions inventory and reported to the European Environment Agency. The quality of this data depends directly on the quality of the monitoring systems installed on site.
The rise of the European sustainability reporting framework through instruments such as the Corporate Sustainability Reporting Directive (CSRD) and the European Sustainability Reporting Standards (ESRS) adds a new dimension: industrial companies must now report their NOx emissions not only to environmental authorities but also to investors, ESG analysts and the market as a whole. In this context, the traceability and credibility of monitoring data become a corporate reputation asset.
Data-driven decision-making
NO monitoring data has value beyond compliance; it forms the basis of truly evidence-based environmental management. In practice, this means:
- In urban environments: local authorities with dense NO monitoring networks can accurately assess the effectiveness of LEZs, justify traffic restrictions to citizens with objective data and evaluate the real impact of public transport fleet renewal.
- In industrial facilities: operators that continuously monitor NO can optimise combustion processes in real time, reducing emissions without sacrificing energy efficiency and anticipating emission peaks that could lead to regulatory non-compliance.
- In environmental policy: air quality improvement plans required by Directive 2008/50/EC (and soon by Directive 2024/2881) must be based on monitoring data that demonstrate both the problem and the impact of the measures adopted. Without sufficiently long and reliable NO and NO2 time series, these plans lack the empirical basis needed to be effective.
Integration with multiparameter sensor networks
The most relevant trend in environmental monitoring in recent years is the integration of NO as one of several parameters in multiparameter continuous monitoring systems. These systems combine, in a single measurement node, sensors for NO, NO2, O3, CO, SO2, particulate matter (PM2.5 and PM10), meteorological parameters (temperature, humidity, pressure, wind) and even VOCs, providing a complete and integrated view of air quality at a single measurement point.
The advantages of this approach are multiple:
- Pollutant correlation: simultaneous NO, NO2 and O3 data allow real-time tracking of the photochemical cycle, validating models and detecting anomalies.
- Source identification: the multiparameter signature of an emission (relative proportions of NO, CO, SO2 and particulate matter) enables more precise identification of the emission source than measuring a single pollutant.
- Deployment efficiency: a single multiparameter node installed at a strategic location provides more information per euro invested than multiple single-parameter sensors deployed without a clear strategy.
- Integration into IoT platforms and digital twins: data from multiparameter networks can directly feed smart environmental management platforms, real-time compliance dashboards and predictive air quality models capable of anticipating pollution episodes before they occur.
In summary, nitric oxide monitoring has evolved from a reactive, occasional measurement (activated only when a problem was suspected) into a strategic component of proactive environmental management, both in urban and industrial contexts. The availability of more accurate, more affordable and more connected measurement technologies than ever before means there is currently no technical or economic justification for managing air quality without real-time NO data.

NO is evident for its ability to reduce visibility. It is responsible for the characteristic brown haze over large cities on calm days.
Frequently asked questions about nitric oxide (NO)
What is nitric oxide and how is it formed in the atmosphere?
Nitric oxide (NO) is an inorganic gas composed of one nitrogen atom and one oxygen atom. At ambient temperature and pressure, it is colourless, odourless and highly reactive. It belongs to the group of nitrogen oxides (NOx), together with nitrogen dioxide (NO2), and is the direct precursor of the latter in the atmosphere.
In the atmosphere, NO is mainly formed through two pathways. The first, and most relevant from an environmental perspective, is high-temperature combustion. When air is heated above 1,200 °C in engines, boilers or turbines, nitrogen (N2) and oxygen (O2) react to produce NO directly from the air, regardless of the fuel used. This is known as thermal NO. The second pathway involves natural processes such as soil microbial activity, atmospheric electrical discharges and wildfires, which contribute diffusely to the global nitrogen cycle.
Once emitted, NO has a very short atmospheric lifetime. In the presence of tropospheric ozone, it rapidly oxidises to NO2, initiating the photochemical cycle responsible for ground-level ozone formation and urban smog. This is why, although European legislation regulates NO2 as a criteria pollutant, effective control of NOx pollution requires acting on NO at the emission source.
What is the difference between nitric oxide (NO) and nitrogen dioxide (NO2)?
Nitric oxide (NO) and nitrogen dioxide (NO2) are two different compounds within the NOx group, with distinct properties and effects. NO is colourless, odourless and generated directly in combustion processes; it is the primary pollutant, the one emitted from stacks or exhaust pipes. NO2 is reddish-brown, has a pungent odour and is far more toxic; it is mainly a secondary pollutant, formed when NO reacts with ozone or oxygen in the air after emission.
This distinction has direct regulatory implications. European legislation regulates NO2 in ambient air (annual limit of 40 µg/m3 under Directive 2008/50/EC), but the origin of the problem lies in NO. Controlling NO at source is the most effective strategy to reduce NO2 in the air we breathe.
What sources generate nitric oxide emissions?
Among human-related sources, the most relevant are road traffic (especially diesel engines, which operate at high temperatures favourable to thermal NO formation) and industrial combustion from power plants, boilers, furnaces and gas turbines. Refineries and the petrochemical sector concentrate multiple emission sources within a single facility, including process heaters, flares and turbines. Maritime transport is a growing source, particularly in port areas and coastal routes.
Natural sources include soil microbial activity (enhanced by the use of nitrogen fertilisers), atmospheric electrical discharges, which directly produce NO, and wildfires, whose smoke can transport NOx over hundreds of kilometres from the original source.
How is nitric oxide measured and monitored in air quality?
The reference method for measuring NO in ambient air is chemiluminescence, defined in the European standard UNE-EN 14211 and the EPA standard 40 CFR Part 50, Appendix F. Its principle is based on the reaction between NO in the sample and internally generated ozone: the emitted light is proportional to the gas concentration, with detection limits as low as 0.4 ppb. It is the method used in official air quality monitoring networks across the EU.
For industrial applications, CEMS systems (continuous emissions monitoring) integrate chemiluminescence analysers or TDLAS laser technology directly into stacks and ducts, reporting hourly averages comparable to permit ELVs.
In urban environments and distributed applications, low-cost electrochemical sensors enable deployment of dense multiparameter networks that complement reference stations with greater spatial coverage, feeding real-time air quality management platforms.
Why is it important to control nitric oxide in urban and industrial environments?
Controlling nitric oxide (NO) is the most effective intervention to improve air quality because it addresses the source of the problem rather than its consequences. In the atmosphere, NO is rapidly converted into NO2 and participates in the formation of tropospheric ozone and photochemical smog, pollutants directly linked to respiratory and cardiovascular diseases and premature deaths.
In urban environments, monitoring NO allows traffic restriction protocols to be activated before NO2 levels reach dangerous thresholds and enables objective evaluation of measures such as Low Emission Zones (LEZs).
In industrial facilities, continuous NO measurement is a legal requirement under the IED Directive and a combustion process optimisation tool. It enables emission reduction at source without compromising energy efficiency, anticipation of regulatory breaches and traceability of data required for environmental reporting and ESG standards.
Conclusion: Nitric oxide, small in size, decisive in environmental impact
Two atoms and a molecule weighing barely 30 g/mol give rise to a colourless, odourless gas, above all imperceptible to the senses. Yet nitric oxide (NO) is one of the most influential compounds in the chemistry of the air we breathe, in the health of millions of people and in the most demanding environmental regulatory frameworks.
Regulation measures NO2, but the problem begins with NO. Acting at the source (emission point, combustion process, industrial facility or urban traffic source) is always more effective, more cost-efficient and more sustainable than managing consequences. And to act at the source, it must be measured at the source.
This is where the strategic importance of real-time nitric oxide monitoring lies. Not as a bureaucratic requirement, but as the operational intelligence that enables industries, cities and public authorities to make data-driven decisions, anticipate pollution episodes, demonstrate regulatory compliance with full traceability and ultimately contribute to healthier environments for the people who live in them.
Next-generation nitric oxide sensors (more accurate, more connected and more easily integrated into multiparameter systems) have removed the technical and economic barriers that for decades limited continuous monitoring to large facilities with substantial budgets. Today, any industrial site, municipality or operator seeking to manage its environmental impact rigorously has the tools to do so.
Indeed, NO is a small molecule. But the challenge of controlling it is not. With the right data, at the right time and in the right place, it is within reach of those willing to address it seriously.




