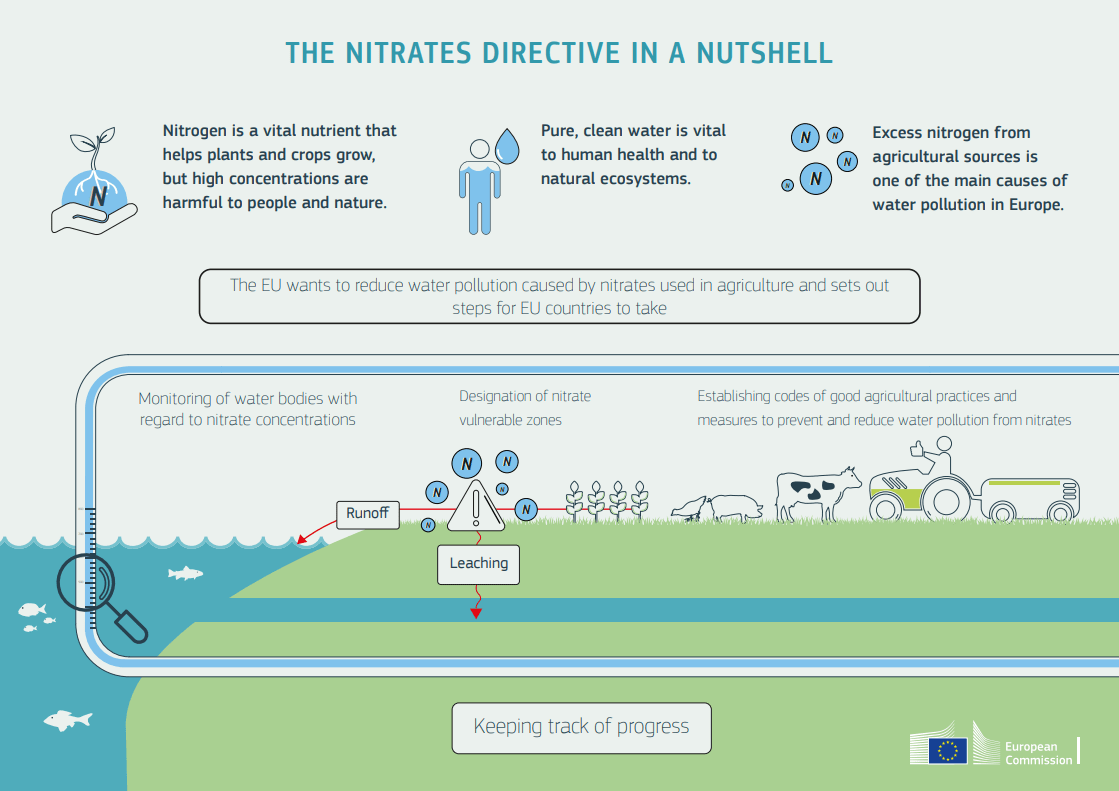
The progress of mankind is largely based on scientific findings. This was the case at the beginning of the last century with the exponential growth in agricultural production.
It became possible when two German chemists devised a method for making liquid ammonia, one of the essential ingredients in the composition of synthetic fertilisers. Nitrogen fixing through an industrial process was an extraordinary agricultural breakthrough to feed a rapidly growing world population.
Liquid ammonia is produced by combining hydrogen with nitrogen, a gas which remains naturally inert in the air in large proportions (78%) and which, however, cannot be directly fixed by plants.
No one could foresee then that an essential element for life on our planet such as nitrogen (a vital component of DNA and proteins of any the planet’s life forms) would be converted into nitrogen compounds by its excessive use in synthetic fertilisers, and which, when returned to the soil, water and air, would make nitrogen harmful to life. It is currently one of the biggest environmental problems facing humanity.
In fact, only 30 to 50% of the synthetic fertiliser employed is used by the plant. The rest of the nitrogen denitrifies, percolates into the soil as nitrates, which then pass into the groundwater and evaporate into the air (Danso & Eskew, 1984).
Air Quality Innovation in Just 1 Click
Stay informed about the air you breathe!
Subscribe to our newsletter to receive the latest updates on environmental monitoring technology, air quality studies, and more.
When they come into contact with the atmosphere, they oxidise, forming the so-called nitrogen oxides (NOx), which are basically nitrogen monoxide and nitrogen dioxide. Together with other gases present in large proportions in the air, such as carbon dioxide and methane, they are major contributors to the ozone-depleting greenhouse effect that drives climate change.
The presence of nitrogen compounds in the air seriously affects public health. In general, it damages the respiratory tract causing various conditions, but it also affects the circulatory and immune systems, and has been shown to be linked to pathologies which develop in organs such as the liver and spleen.
In order to prevent damage to health being further aggravated by synthetic fertiliser use and thus adding polluting gases to the air, such as nitrogen oxides, early detection is crucial to enable an alert to be given in the event of any of these gases exceeding pre-established values; this will help efficient decision-making by managers. There is also a need to control the fertiliser industry’s environmental impact.
The fertiliser industry’s impact on air quality
For farmers to achieve higher and improved crop production, the synthetic fertiliser manufacturing industry needs to control and anticipate, according to the regulations established by law, the environmental risks caused by chemical processes (acid dissolution, pressure, high temperatures, etc.) used during their production.
During the manufacturing process, soot and dust particles are normally emitted into the air along with polluting gases such as sulphur oxide (SOx), ammonia (NH3)Invisible yet powerful: ammonia (NH3) is a colourless gas which, although naturally present in the atmosphere in small amounts, can become an unwelcome ene...
Read more, and nitrogenous by-products such as nitric oxide (NO)Nitric oxide (NO) is one of the most important, and often underestimated, gases in the field of air qualityAir quality refers to the state of the air we breathe and its composition in terms of pollutants present in the atmosphere. It is considered good when poll...
Read more and industrial emissions management. Colourless...
Read more and nitrogen dioxide (NO2)Nitrogen dioxide (NO2) is a harmful gas whose presence in the atmosphere is mainly due to the use of fossil fuels in combustion vehicles and industrial act...
Read more in addition to emissions of highly polluting volatile solvents. All of them directly affect the environment as well as human health.

Agricultural field fertilisation

Looking to control the impact of your processes on air quality?
Download the technical report [PDF] on the air quality monitoring network deployed at the Cemex plant.
Types of fertilisers
Agricultural fertilisers supplement the vegetative development of crops by providing those nutrients that are scarcest in the soil, thus encouraging plants to grow and reproduce adequately.
Its action is based on the supply of three essential chemical elements: nitrogen, phosphorus and potassium. Other micronutrients such as zinc, iron, manganese, copper, etc. are added, which, in turn, improve yields and promote the health of the plants being cultivated.
A distinction is made depending on the origin of the nutrients on which the composition of the fertiliser is based:
Organic fertilisers
Manure, obtained from domestic animal excrement and plant remains, has traditionally been used as a source of carbonaceous material to stimulate crop development. This happens because they are broken down by soil micro-organisms and converted into mineral nutrients that can be assimilated by plants as they are gradually incorporated. This addition of organic matter helps to prevent soil fertility loss. Organic matter in turn contributes to the soil’s porosity and permeability, essential physical factors for adequate water retention. This type of fertiliser is the most commonly used in organic farming.
Synthetic or inorganic fertilisers
These are also called chemical fertilisers because they are based on industrial chemical reactions that break down inorganic elements such as salts, gases and rocks. They are the most widely used fertilisers in agriculture because they help make nutrients available to plants more quickly. They improve crops because they are easily absorbed by plants and have an impact on increasing yields. Their effective use, which does not generate surpluses that pollute the environment, as well as technological advances in the fertiliser industry are minimising the toxic effects that this type of chemical fertiliser generates on public health and safety, as well as on the natural environment’s fauna and flora.
Biofertilisers
This is the name given to fertilisers that use living micro-organisms to nourish plants. They stimulate biological processes in crops and improve their nutrient uptake efficiency. They do not generate harmful surpluses that remain in the soil or pollute the water or air and are therefore widely used in organic farming.
Organic-mineral fertilisers
These have a mineral origin which is supplemented during their chemical manufacture by mixing with organic nutrients from animal or vegetable carbonaceous materials. In addition to supporting plant nutrition, they contribute to improving soil properties.
Types of pollution caused by fertilisers
While the fertiliser industry is essential to modern agriculture, sustaining the world’s food supply, the overuse of synthetic fertilisers is having a serious environmental impact.
Nitrogen released through synthetic fertilisers returns to the environment in the form of nitrogen oxides that affect the:
Atmosphere
Excess nitrogen returns to the atmosphere in the form of nitrogen oxides whose contribution to the greenhouse effect is 300 times more harmful than that caused by carbon dioxide.
Soil
At the same time, excessive fertiliser use also causes nitrogen to remain in the soil, affecting its microbiology while altering the ratio of carbon and phosphorus. Soil eutrophication due to excess nutrients and even acidification of agricultural land; leading to loss of fertility and imbalance in the physico-chemical properties of the soil structure (Pahalvi et al., 2021).
Water
In addition, through run-off and leaching from agricultural soils, nitrogen is incorporated as nitrites and nitrates into groundwater, inland water bodies such as lakes and ponds, as well as into the sea.
In this way it enters the water cycle, contaminating water sources for various human uses. This is a serious environmental problem that in turn affects biodiversity by altering the natural environment of many species of flora and fauna that depend on water for their survival.
Its direct impact on aquatic ecosystems leads to eutrophication, which alters water conditions by blocking the passage of light and reducing dissolved oxygen, seriously deteriorating the conservation status of water-related flora and fauna.
Health problems arising from fertiliser use
While nitrogen is essential for the growth of agricultural crops, its overuse through fertilisers causes damage to both health and the environment.
Every year, around seven million people die prematurely, with an annual economic impact of €8 trillion from diseases developing as a result of the air pollution we breathe, both indoors and outdoors.
Among the polluting gases that remain in the atmosphere, nitrogen oxides from the use of inorganic fertilisers account for a significant proportion. Breathing them in causes serious damage to human health, such as respiratory diseases, circulatory system disorders, or damage to organs such as the liver and spleen.
In countries considered the breadbasket of the world, such as India, industrialisation of agriculture, whilst mitigating its population’s starvation, has been a source of endocrine and neurotoxic disorders, respiratory diseases, and bladder, ovarian and lymphatic cancers are all highly prevalent.

Farmers in cultivated fields
In the Punjab state of India, where huge areas are dedicated to agriculture, the declining health of farmers and their families is directly related to the use of fertilisers and pesticides. Their use increases as the soil’s fertility is depleted, as its water and nutrient holding capacity decreases, requiring greater amounts of synthetic fertilisers to be used to grow crops.
“Thirty years ago, we only fertilised the fields with sulphate twice a year. Now we use chemicals four or five times a year”
Rapid action to tackle this serious air pollution problem affecting mankind’s health requires that the causes of ozone depletion be reduced by continuous monitoring and regulation of the presence and proportion of nitrogen oxides in the air.
While much of gas pollution comes from vehicle combustion and the energy industry, modern farms using a large amount of synthetic fertilisers and pesticides also make a significant contribution.
Strategies for reducing the use of agricultural fertilisers
Preventing the release of excess nitrogen from synthetic fertilisers into the atmosphere is one of the great challenges for both science and industrial research and for the adequacy of regulations on their use.
Firstly, accurate measurements of agricultural emissions must be obtained without stopping the basic production of the food needed to feed the world. Policies must also balance the social and political spheres to minimise impact on the atmosphere as it affects both the environment and human health (Aneja et al., 2009).
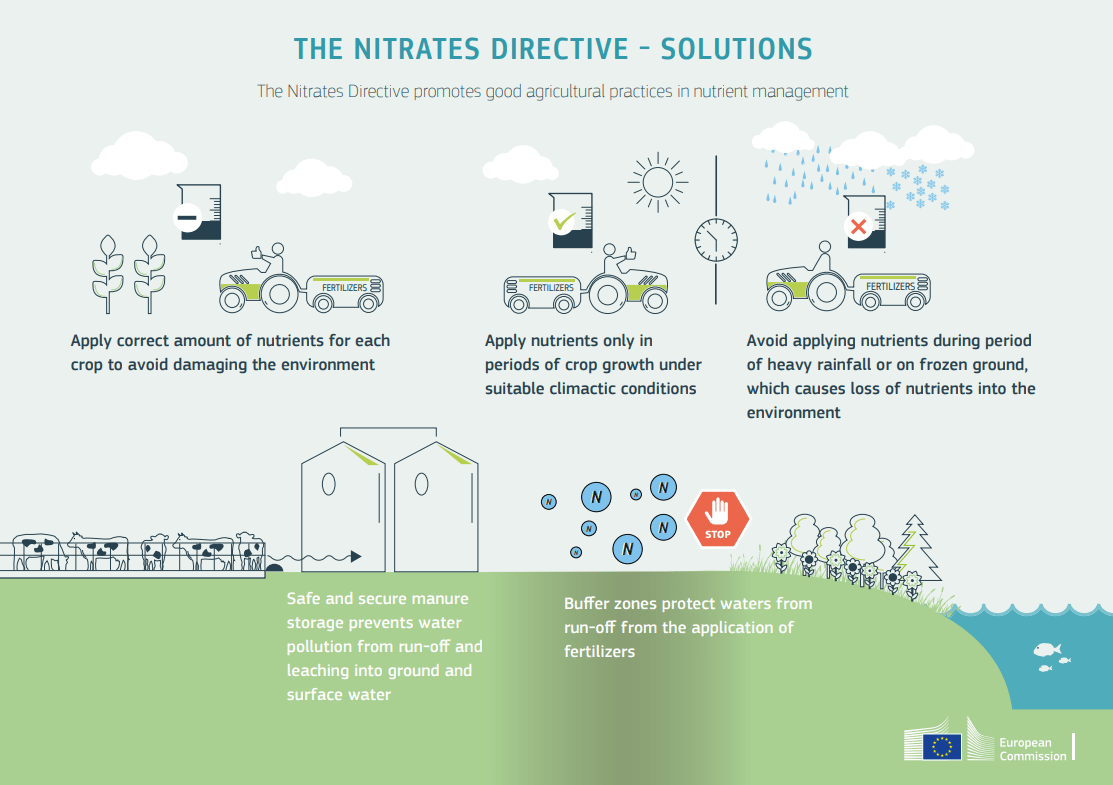
Among the measures to be taken is the use of fertilisers in suitable proportions and composition for the needs of each type of crop. Furthermore, nutrients should only be applied under appropriate environmental conditions so that they only affect the vegetative growth of crops and do not cause losses of fertilisers which will end up in the environment.
It is necessary to implement the use of organic and mineral fertilisers as they provide nutrients to plants and improve agricultural crops in a more balanced way for the soil, and without emitting synthetic derivatives that pollute soil, water and air without affecting natural diversity.
This means encouraging crops free of fertiliser residues such as nitrates through sustainable agricultural practices such as crop rotation and organic farming. Techniques that have emerged in modern agricultural activities as an effective way to provide high added value to products and, at the same time, mitigate the impact of chemical fertilisers on human health and the environment.
Apart from promoting the efficient use of chemical fertilisers, the natural process of biological fixation of molecular nitrogen (N2) must be improved. This was the basis of agriculture before synthetic fertiliser manufacturing began.
While no plant can fix molecular nitrogen biologically by itself, symbiosis with micro-organisms, which break down the stable nitrogen molecule and then form ammonia and nitrates, can be achieved in some crops. Although this is not the case for wheat, rice and maize, the staple foods of most of humanity. Other cultivated plants such as peas, beans and lentils are capable of biological nitrogen fixation with the help of bacteria.
A revolutionary technology is currently being researched (N-Fix) that allows plants to take nitrogen directly from the air and convert it into ammonia whilst respecting the environment. N-Fix technology has achieved this by stimulating plants with the introduction of nitrogen-fixing bacteria into the roots of most cultivated plants.
Based on imitating nature, permaculture seeks to make crops sustainable and at the same time cause zero environmental risk by promoting agriculture that imitates natural processes.
Thus, agroforestry integrates trees (forestry) with cultivated plants and livestock activity so that, by growing together, they obtain mutual benefits in terms of nutrients and availability of multiple resources. Polycultures or mixed crops encourage different plants to share the same space, promoting biodiversity, optimising resources, improving soil fertility and preventing pests and diseases. Terrace, row or spiral cultivation have also proven to be beneficial both for farmers and the environment whilst improving soil composition and water optimisation by improving soil drainage.
There are many ways to achieve agriculture that preserves the environment and contributes to climate action. Responsible production, which is necessary for the health and well-being of the planet’s living beings, is essential for achieving food security, as well as for not altering the quality of the air we breathe.
Chemical fertilisers versus organic fertilisers
For a transition towards more respectful and sustainable agricultural practices concerning the health of the planet and its inhabitants, the use of organic or ecological fertilisers should be encouraged. By not providing nitrogen through a synthetic compound such as liquid ammonia, they are not harmful and stand out as a crucial alternative to tackle the problematic pollution caused by synthetic fertilisers.
Organic fertilisers, being composed of carbonaceous material, can be absorbed more gradually by plants. Soil micro-organisms must act by breaking them down before they can be used by the plant. However, such a natural process ensures the availability of supplementary nutrients in the soil for a longer period.
However, in the case of synthetic fertilisers, nutrients are immediately available to plants, which favours rapid incorporation into the plant’s vascular system and, in the same way, leading to a substantial improvement in crop growth.
Organic fertilisers that add mineral compounds from salts and rocks are also more environmentally sustainable because they do not require chemical processing. However, although they are environmentally friendly and nourish plants, nitrogen is present in their composition, although in a lower proportion than in synthetic fertilisers.

A tractor spreading fertiliser on a crop
Measuring environmental impact with Kunak AIR sensors
Synthetic fertilisers affect the atmosphere by releasing reactive nitrogen which oxidises into nitrogen monoxide and nitrogen dioxide (NOx). This process leads to several environmental issues, including the fact that these gases are extremely damaging to the ozone layer (O3). Their destruction encourages the greenhouse effect that accelerates global warming.
With agricultural practices generating 60-70% of nitrous oxide emissions into the air through the use of chemical fertilisers, it is crucial to promote the use of less polluting fertilisers for the atmosphere, as well as to have accurate and adequate data to estimate the presence and proportion of nitrogen oxides (nitrogen dioxide and nitrogen monoxide) in the atmosphere and to monitor ozone levels.
The synthetic fertiliser industry can mitigate its polluting emissions which alter air quality by installing a perimeter ring of sensors to monitor the emissions of polluting gases. This will avoid exceeding the legally established limits.
Such monitoring would, in turn, allow early detection and activation of warning systems in the event of anomalies such as the leakage of polluting gases caused by problems or anomalies during the production process, thus avoiding incurring extra costs in maintenance and repairs.
Although environmental factors such as humidity, temperature and soil type affect the nitrogen cycle in agriculture and thus the polluting gases emitted into the atmosphere by the use of synthetic fertilisers, continuous monitoring of the gases is recommended.
The deployment of a network of solar-powered Kunak AIR stations in agricultural areas will detect the presence of nitrogen compounds in the air when synthetic fertilisers are applied to plants.
It is also desirable to carry out real-time monitoring to detect critical moments during plant growth that involve the release of nitrogen into the air, which will eventually become emissions that alter air quality due to the nitrogen surpluses emitted.
Similarly, to protect human health and the environment, it is necessary to have a perimeter monitoring network in fertiliser production plants to control other pollutants derived from the production process such as sulphur oxide (SOx), ammonia (NH3) and nitrogenous by-products such as nitric oxide (NO) and nitrogen dioxide (NO2), in addition to emissions of high polluting volatile solvents.
The analysis of the data collected by the sensor network through tools, such as the Kunak AIR Cloud software, allows the levels of those pollutants that have the most significant impact on air quality to be exhaustively controlled, thereby promoting sustainability in agriculture.
Sources and references
European legislation on fertilisers
Aneja, V. P., Schlesinger, W. H., & Erisman, J. W. Effects of agriculture upon the air quality and climate: research, policy, and regulations. Environmental Science & Technology, 2009 43 (12), 4234-4240.
Pahalvi, H. N., Rafiya, L., Rashid, S., Nisar, B., & Kamili, A. N. (2021). Chemical fertilizers and their impact on soil health. Microbiota and Biofertilizers, Vol 2: Ecofriendly Tools for Reclamation of Degraded Soil Environs, 1-20.
Danso, K. A., & Eskew, D. L. (1984). Nitrogen fixation in fababeans as affected by plant population density in sole or intercropped systems with barley. Soil Biology and Biochemistry 19.4 (1987): 411-415.